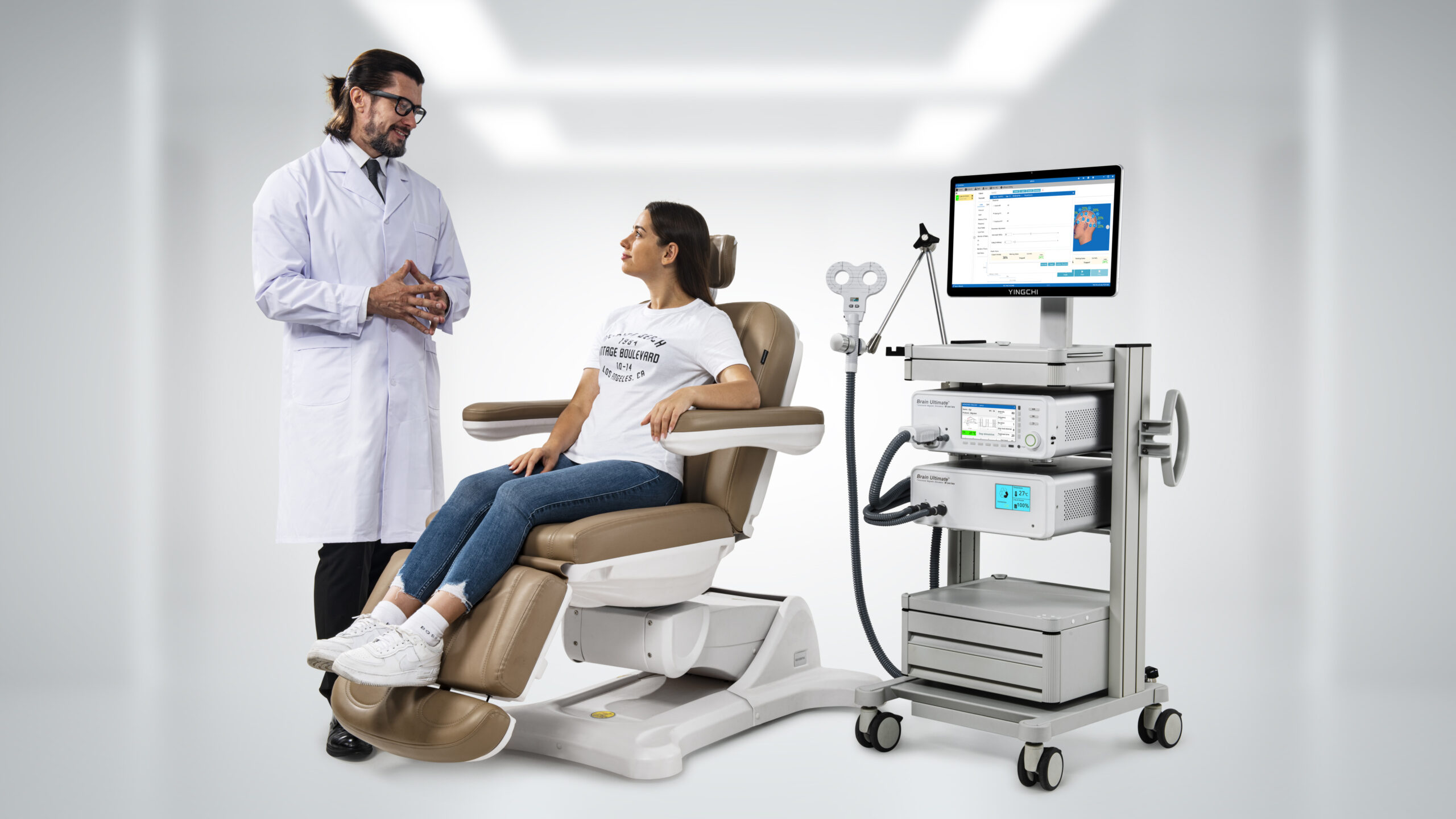
Brain Ultimate TMS M-Series
The Most Advanced Affordable TMS System
Experience the only TMS device that uses pure deionized water for true 24/7 operation. While others rely on transformer oil and complex cooling systems, our proprietary design delivers continuous therapy with unmatched safety, simplicity, and no per-pulse fees.
Brain Ultimate TMS by the Numbers
Introduction to Transcranial Magnetic Stimulation (TMS)
Transcranial Magnetic Stimulation (TMS) is a non-invasive method of brain stimulation. It works by directing a magnetic field pulse through the skull, where it induces electrical currents in the underlying brain tissue through Faraday's law of electromagnetic induction. TMS can both depolarize and hyperpolarize cortical neurons, influencing the immediate stimulation area and connected neural structures.
Beyond the direct stimulation site, TMS influences cortical and subcortical structures connected synaptically. The neurophysiological changes induced by repetitive TMS can persist beyond the stimulation period through long-term potentiation and depression mechanisms — making TMS an effective FDA-cleared treatment for Major Depressive Disorder, Obsessive-Compulsive Disorder, and additional indications.
M-Series Ultimate TMS Device Features
Our TMS systems adhere to international safety standards with certifications including FDA, CE, CFDA, TGA, KFDA, and NMPA.
Advanced Safety Features
Comprehensive protection systems ensure patient and operator safety during every treatment session.
- Dual-tier temperature protection: 41°C warning, 42°C auto-stop
- Continuous monitoring of coolant level, temperature, and circulation
- Enhanced patient data security with dongle authorization
- Real-time system status indicators
Precision Positioning System
Advanced positioning technology ensures accurate and repeatable coil placement for optimal treatment outcomes.
- 10-20 system positioning caps (S/M/L) for defined brain regions
- Built-in coil position maps in QuickStim software
- Flexible coil arm for precise adjustments
- Compatible with neuronavigation systems
User-Friendly Software
QuickStim software provides comprehensive control with an intuitive touchscreen interface designed for clinical efficiency.
- Effortless patient file creation and management
- Built-in FDA-cleared protocols and custom protocol creation
- Hot key access for frequently used protocols
- Cloud connectivity from approved devices with QuickStim
Clinical Versatility
FDA-cleared for multiple conditions with four distinct treatment protocols to meet diverse patient needs.
- Standard MDD Protocol (19 minutes)
- Enhanced MDD Protocol (37 minutes)
- iTBS Protocol (3 minutes)
- OCD Protocol (18 minutes)
Practice Enhancement
Maximize clinic efficiency and patient throughput with no per-pulse or per-use fees.
- True 24/7 operation with deionized water cooling
- 20,000,000 pulse coil lifespan
- Lightest coil on the market for operator comfort
- Comprehensive installation, training, and ongoing support
Integration & Compatibility
Seamlessly integrates with existing clinical workflows and diagnostic equipment.
- Compatible with EEG monitoring systems
- MEP Module integration ($2,950 add-on)
- Neuronavigation system compatible
- Cloud-connected treatment management
The Only TMS with Pure Deionized Water Cooling
While other systems rely on transformer oil and complex cooling mechanisms, our proprietary technology uses only medical-grade deionized water for unmatched safety and simplicity.
Pure Deionized Water
Unlike competitors using transformer oil, our system uses only medical-grade deionized water — eliminating the risks associated with oil-based cooling and ensuring a cleaner, safer treatment environment.
True 24/7 Operation
Continuous operation without overheating or performance degradation. Your clinic can treat patients all day without cooling breaks or system downtime.
Closed-Loop Safety
Sealed circulation system prevents leaks and contamination while maintaining optimal cooling efficiency with real-time temperature monitoring.
Exceptional Durability
Most clinics operate 2–3 years between coolant service. Resistant to electrolysis, corrosion, and blockages. Refill at 85% capacity — no complex maintenance required.

Comprehensive Specifications
QuickStim Software Platform
Comprehensive software designed for clinical efficiency with cloud connectivity from approved devices.
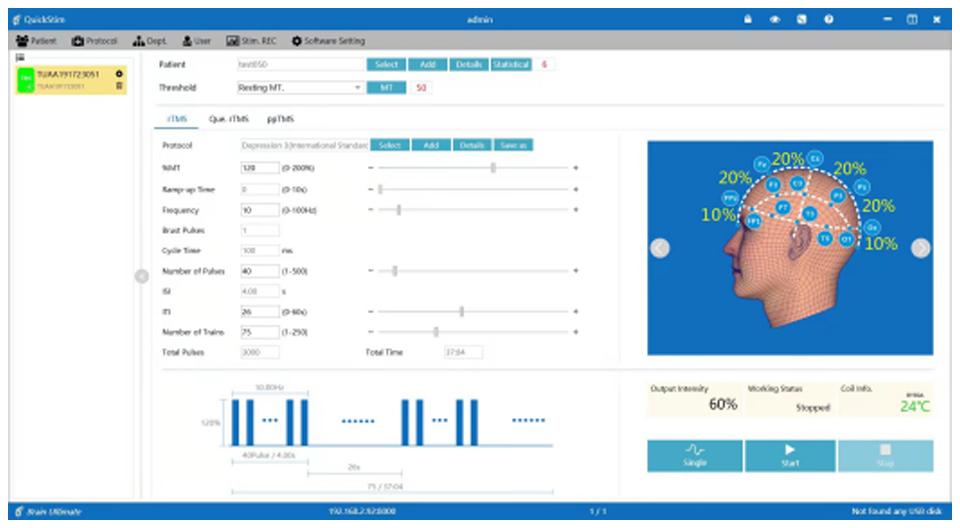
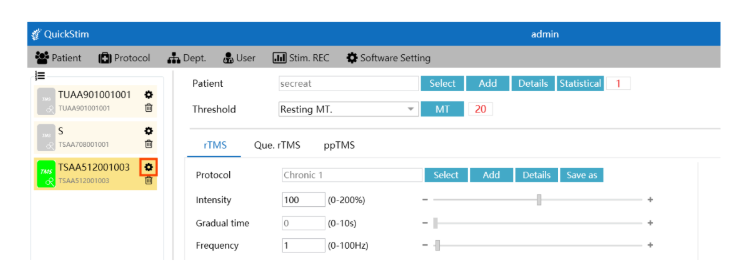
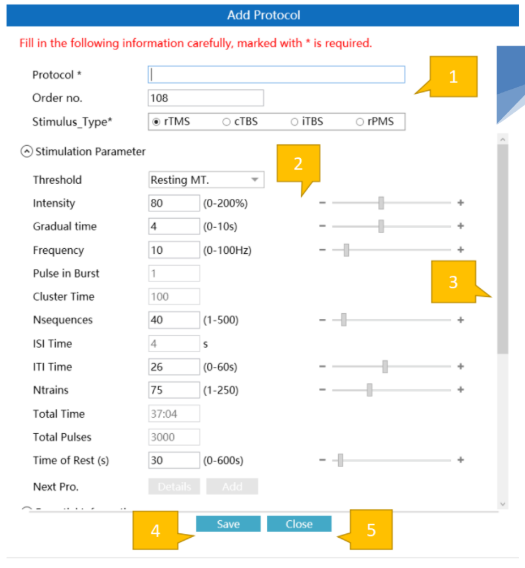
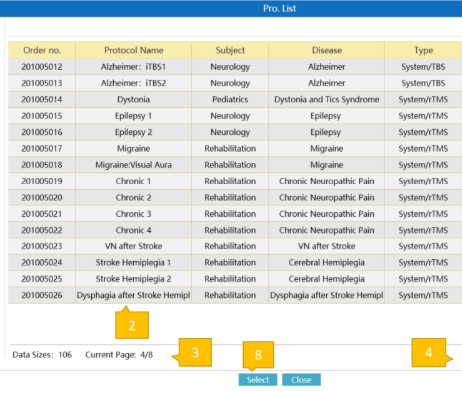
Streamlined Clinical Workflow
QuickStim software simplifies every aspect of TMS treatment delivery, from patient intake to treatment completion and reporting.
- Effortless Patient ManagementQuick patient file creation, comprehensive history tracking, and secure data storage with dongle authorization.
- Built-in FDA-Cleared ProtocolsPre-loaded with all 4 FDA-cleared protocols plus custom protocol creation. Hot key access for frequent settings.
- Cloud ConnectivityAccess patient data across devices with Brain Ultimate Cloud integration — from approved devices where QuickStim is installed.
- Comprehensive ReportingGenerate treatment summaries, progress reports, and session documentation for clinical records.
Experience Our Technology Firsthand
Join the 50+ clinics nationwide that trust Brain Ultimate for reliable, affordable, and advanced TMS therapy — backed by 176 peer-reviewed studies and no per-pulse fees.
